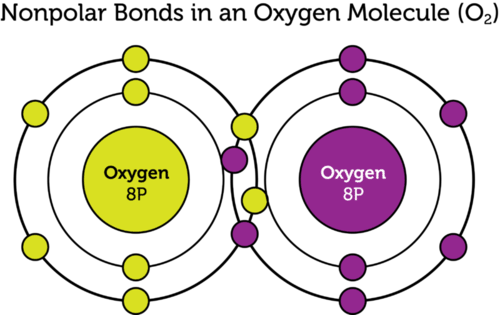
Three Examples Of Nonpolar Covalent Bonds
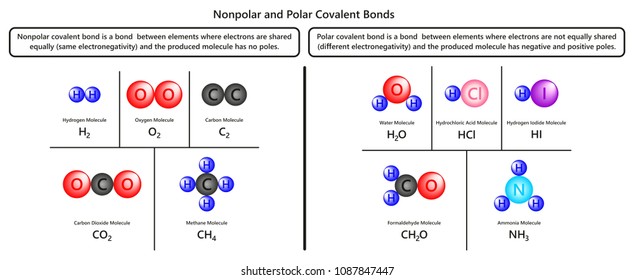
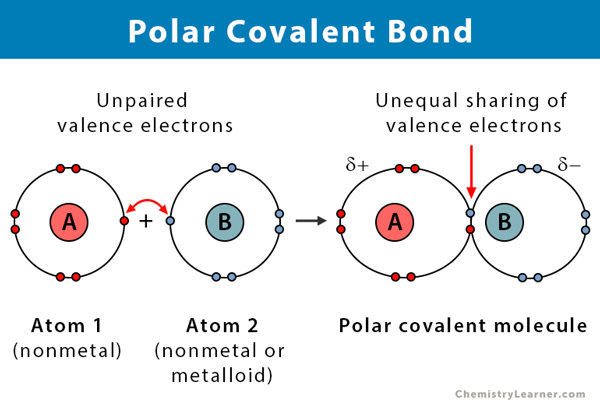
The carbon atom has four electrons in its outermost shell and needs four more. Polar Covalent Bond When the electrons spend more time around the more non-metallic atom the sharing of the electron pair becomes unequal and results in the formation of polar covalent bonds.

Covalent Bonds Biology For Majors I
An unsaturated fatty acid occurs when a double covalent bond replaces a single covalent bond and two hydrogen atoms Figure 2.
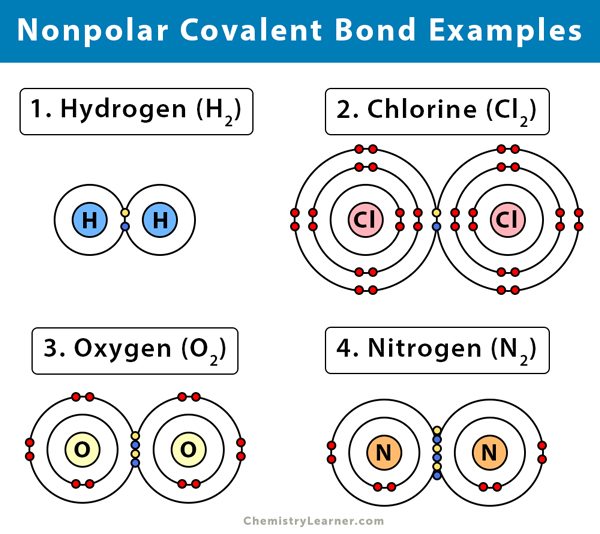
Three examples of nonpolar covalent bonds. If you know the chemical formula of a compound you can predict whether it contains ionic bonds covalent bonds or a mixture of bond types. A covalent bond with an equal share of electrons and an electronegativity difference of zero is called a nonpolar covalent bond. A bond in which the electronegativity difference is less than 19 is considered to be mostly covalent in character.
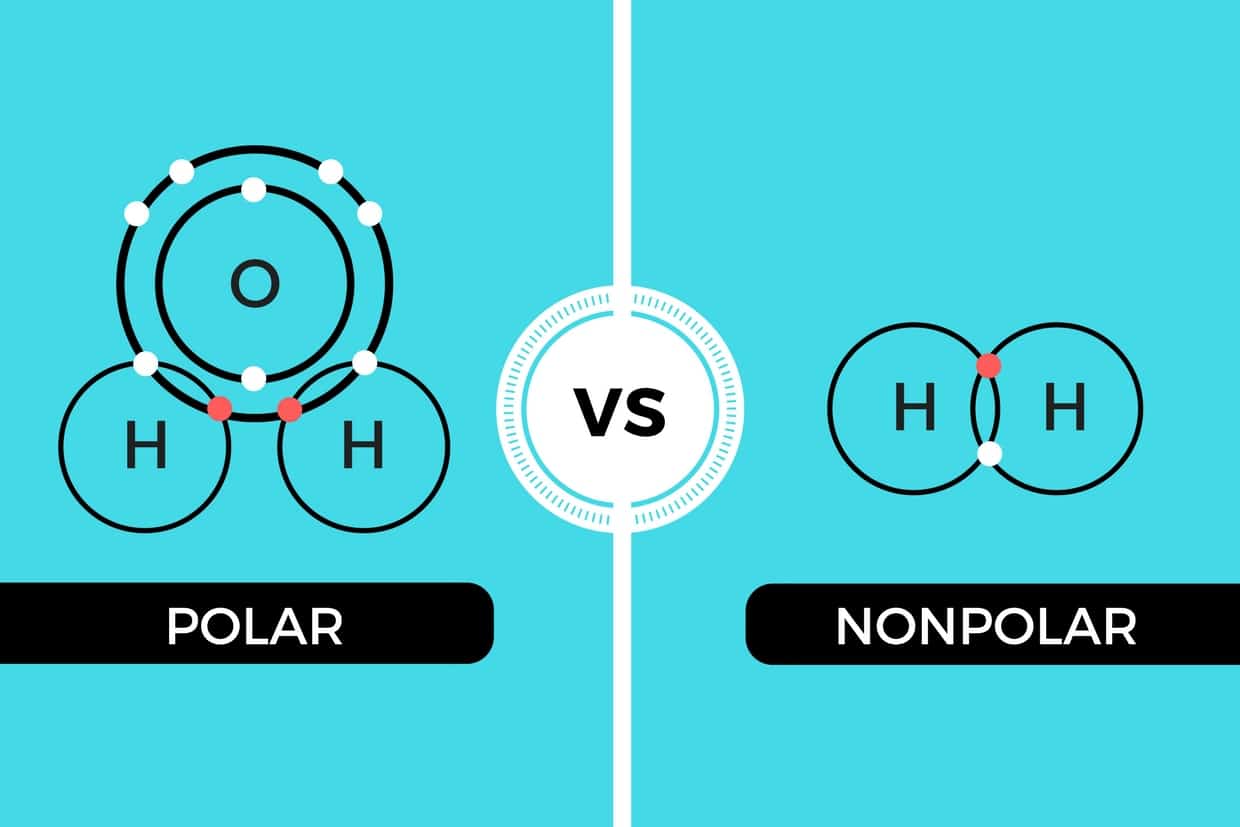
Nonpolar molecule are water fearing and do not easily dissolve in water. In some cases three covalent bonds can be formed between two atoms. Covalent bonds between identical atoms as in H 2 are nonpolarie electrically uniformwhile those between unlike atoms are polarie one atom is slightly negatively charged and the other is slightly positively charged.
Its considered to be a polar covalent bond and if your difference in electronegativity is 04 thats considered to be nonpolar covalent bonds so somewhere in between there must be the difference between nonpolar covalent bond and a polar covalent bond and most textbooks will tell you. It is very important to understand that although PCl5 has polar bonds within it it is a nonpolar in nature due to its symmetrical shape. The most common gas in the atmosphere nitrogen is made of two nitrogen atoms bonded by a triple bond.
Another example of a nonpolar covalent bond is found in the methane CH 4 molecule. Examples of such compounds. In the formation of a nitrogen molecule each nitrogen atoms having five valence electrons provides three electrons to form three electron pairs for sharing.
Molecules containing polar bonds have no molecular polarity if the bond. Polar molecules must contain one or more polar bonds due to a difference in electronegativity between the bonded atoms. Covalent molecular structures are compounds containing molecules with covalent bonds.
A The electrons in the covalent bond are equally shared by both hydrogen atoms. This partial ionic character of covalent bonds increases with the difference in the electronegativities of the two. Triple covalent bonds are represented by three dashes and are the least stable types of covalent bonds.
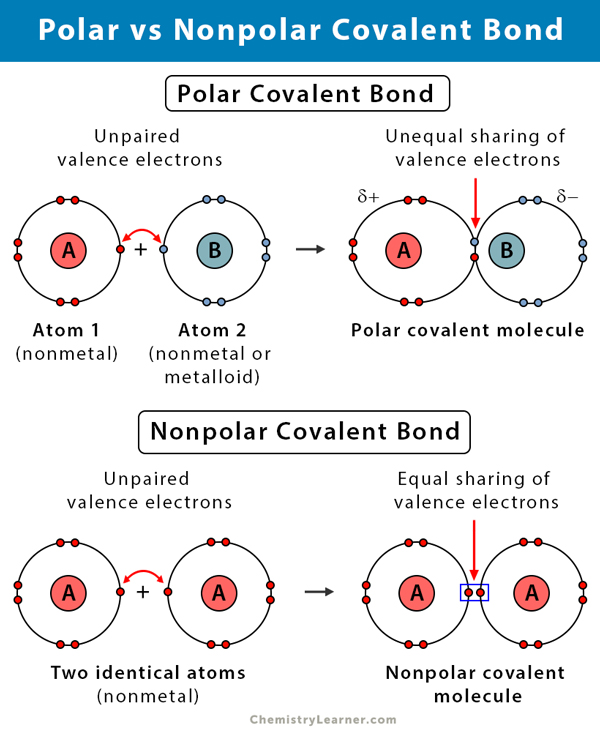
A covalent bond that has an equal sharing of electrons and the electronegativity difference is zero is called a nonpolar covalent bond. Sharing of electrons allows the atoms to obtain the stable outer electron shell configuration. Thus as the electronegativity difference of the two bonded elements increases a nonpolar bond gives way to a polar bond which in turn becomes an ionic bond.
Dipole moments are generally found in Polar Covalent Bonds. How an atoms electronegativity ranks on a scale is how the three different bonding types ionic polar covalent and nonpolar covalent are classified. The three bonds lie in a single plane in such a way that three P-Cl bonds make.
While the three B F covalent bonds are formed due to the sharing of electron pairs resulting from contributions of both boron and fluorine atoms an N B bond is formed due to the donation of a lone pair of. Phospholipids look just like lipids except that one of the fatty acid chains is. The Pauling scale measures electronegativity on a scale of 000-400 with 000.
Classifying bonds as covalent polar covalent or ionic. Nonmetals bond to each other via covalent bonds while oppositely charged ions such as metals and nonmetals form ionic bondsCompounds which contain polyatomic ions may have both ionic and covalent bonds. A nonpolar covalent bond is a covalent bond in which the bonding electrons are shared equally between the two.
There are five chlorine atoms attached to the phosphorus and all bonds are covalent in nature. The Octet Rule requires all atoms in a molecule to have 8 valence electrons--either by sharing losing or gaining electrons--to become stable. It requires 8 electrons because that is the amount of electrons needed to fill a s- and p- orbital electron configuration.
This scale is called the Pauling scale. Hydro- means water and -phobic means fear. A non-covalent interaction differs from a covalent bond in that it does not involve the sharing of electrons but rather involves more dispersed variations of electromagnetic interactions between molecules or within a molecule.
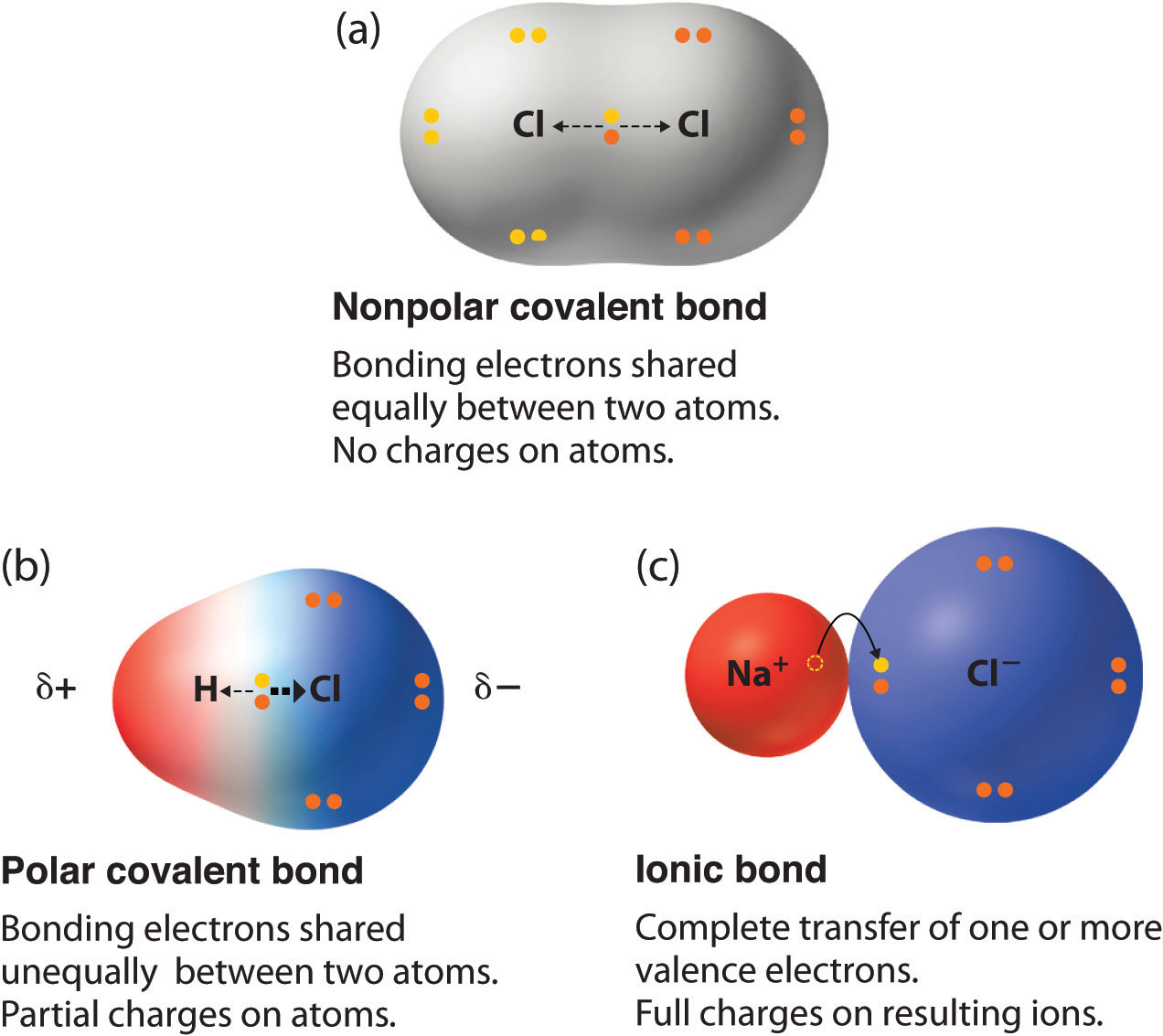
Using the periodic table of electronegativities from the last page write down examples of atom pairs which you would expect to form covalent bonds polar covalent bonds and ionic bonds. Ionic bonds result from a transfer of electrons from one species usually a metal to another usually a nonmetal or polyatomic ion. A covalent bond that has an equal sharing of electrons part a of Figure 45 is called a nonpolar covalent bond.
Nitrogen atoms will form three covalent bonds also called triple covalent between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell. Tics of BOTH ionic and covalent bonding. Covalent bonds result from a sharing of electrons by two or more atoms usually nonmetals.
There are in fact no purely ionic bonds just as there are no purely covalent bonds. Nitrogen N2 hydrogen H2 water H2O ammonia NH3 chlorine Cl2 fluorine F2 are some of the examples of the compounds having covalent bonds. Also known as a.
The chemical energy released in the formation of non-covalent interactions is typically on the order of 15 kcalmol 10005000 calories per 602 10 23 molecules. Technically nonpolar bonding only occurs when the atoms are identical to each other eg H 2 gas but chemists consider any bond between atoms with a difference in electronegativity less than 04 to be a. Bonding is a continuum of types.
However at this point we need to distinguish between two general types of covalent bonds. Electronega-tivity differences determine the balance of character. These molecules have nonpolar covalent bonds or polar covalent bonds both of which share.
This is the main difference between covalent molecular and covalent network. For Covalent bonds atoms tend to share their electrons with each other to satisfy the Octet Rule. The bonding electrons in polar covalent bonds are not shared equally and a bond moment results.
A covalent bond with an unequal sharing of electrons and the electronegativity difference within the range of 01-2 is called a polar covalent bond. Lewis theory Gilbert Newton Lewis 1875-1946 focuses on the valence electrons since. Nonpolar molecules are hydrophobic.
When elements combine there are two types of bonds that may form between them. Polar bonds are the dividing line between pure covalent bonding and pure ionic bondingPure covalent bonds nonpolar covalent bonds share electron pairs equally between atoms. Each nitrogen atom is able to share three electrons for a total of.
In chemistry polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment with a negatively charged end and a positively charged end. For example tetrachloro-methane carbon tetrachloride CCl 4 has polar CCl bonds but the tetrahedral arrangement of the four bonds about the central carbon atom causes the individual bond moments to cancel. Covalent network structures are compounds composed of a network structure with covalent bonds between atoms throughout the material.
However a molecule may be polar or nonpolar depending on its geometry. Polyunsaturated fatty acids have many of these double bonds. Figure 2 distinguishes the covalent bonds from the coordinate covalent bond in NH 3 BF 3.
Figure 45 Polar versus Nonpolar Covalent Bonds. Nonpolar Covalent Bonds.
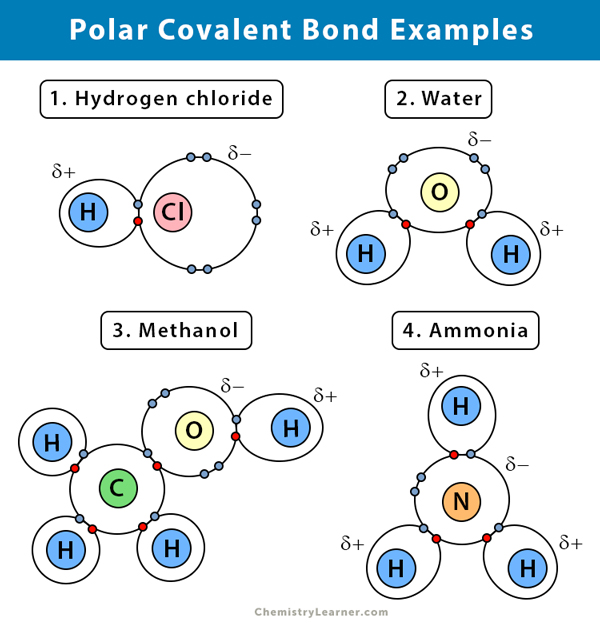
Polar Covalent Bond Definition And Examples

Bond Polarity Chemistry For Non Majors
Nonpolar Covalent Bond Definition And Examples
Difference Between Non Polar And Polar Covalent Bonds Difference Between
Nonpolar Images Stock Photos Vectors Shutterstock
Polar Covalent Bond Definition And Examples

Ionic Bonds Vs Covalent Bonds Chemtalk
Polar Covalent Bond Definition And Examples
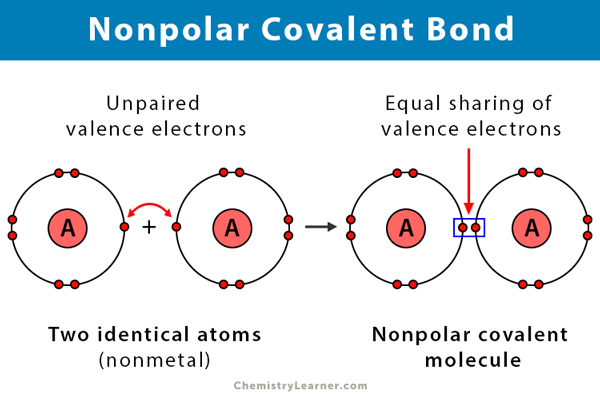
Nonpolar Covalent Bond Definition And Examples
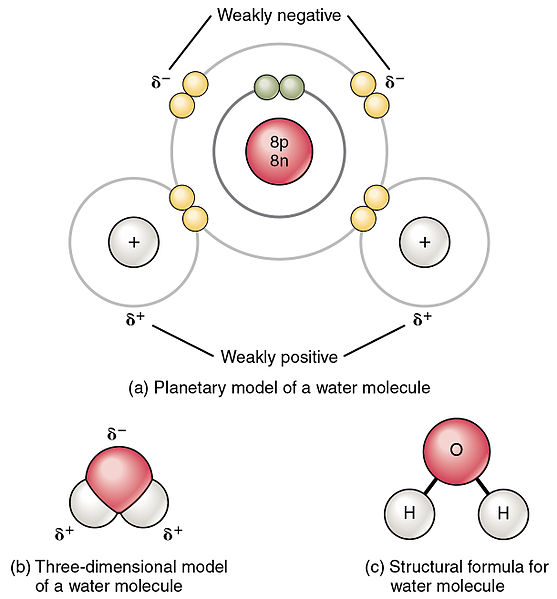
Polar And Non Polar Molecules Vce Chemistry

Polar And Nonpolar Covalent Bonds Definitions Molecules And Examples

4 2 Polar And Non Polar Covalent Bonds Sl Youtube

Polar And Nonpolar Covalent Bonds Overview Examples Expii
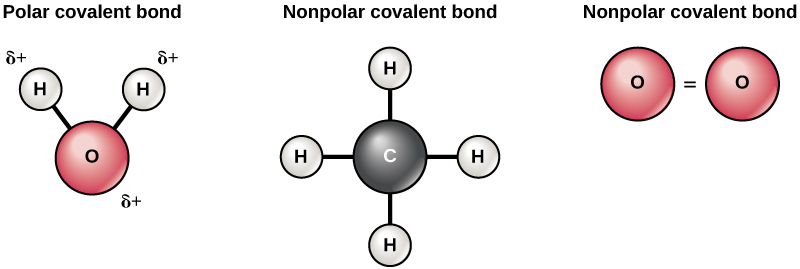
0 1 Chemistry The Building Blocks Of Molecules Gpc By Openstax Page 5 29 Jobilize

Polar Vs Nonpolar It S All About Sharing On An Atomic Level

Chemical Bonds Mhcc Biology 112 Biology For Health Professions
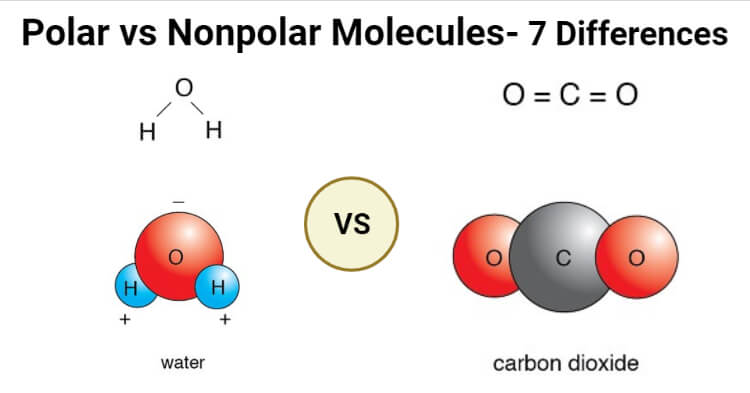
Polar Vs Nonpolar Molecules Definition 7 Key Differences Examples